Formosa Pharmaceuticals Pipeline
Pipeline Overview
Double A R&D Strategy

Our “Double A” strategy focuses on two core pillars: APNT® and ADC.
- APNT® Nanoparticle Formulation Technology: The core advantage lies in solving drug solubility issues to enhance absorption rates. Our flagship ophthalmic drug, APP13007, has successfully obtained FDA approval. This technology is now being extended to inhalation modes and local injections, driving further co-development projects.
- ADC (Antibody-Drug Conjugates): Through a strategic alliance with EirGenix and Formosa Laboratories, we are co-developing TSY-110 and TSY-120, two high-demand HER2 ADC biosimilars. Furthermore, we are developing TSY-310, a novel bispecific ADC targeting solid tumors, actively positioning ourselves in the high-value oncology market.
APNT® Programs
APNT® utilizes salts and/or sugars as milling media to reduce the particle size of poorly soluble drugs to the nanoscale under mild conditions. This process not only minimizes cross-contamination and maintains drug purity but also significantly improves absorption rates and bioavailability.
APNT® has broad applications, with suitable dosage forms covering eye drops, oral formulations, inhalants, and local injections. The successful US FDA approval and market launch of our flagship ophthalmic project, APP13007, clearly demonstrates the high “druggability” of the APNT® platform and its international recognition. Please refer to our R&D projects to learn more.

For Inflammation and Pain after Ocular Surgery Treatment| APP13007
APP13007 is a potent steroid nanoparticulate suspension derived from APNT® for the treatment of post-operative inflammation and pain. Clinical trials have confirmed its high efficacy and low side-effect profile; specifically, the risk of intraocular pressure (IOP) elevation is lower than other steroid eye drops. With a convenient dosage of just one drop, twice daily, it significantly improves patient compliance. Status: Successfully obtained US FDA approval, launched, obtained Health Canada approval, and licensed in multiple global markets.
Infectious Eye diseases Treatment|APP13002

APP13002 is a nanoparticulate suspension eye drop developed with APNT® featuring dual broad-spectrum antibacterial and anti-inflammatory effects. It shows strong potential for treating bacterial conjunctivitis, keratitis, blepharitis, and Meibomian Gland Dysfunction (MGD).
APNT Co-developments
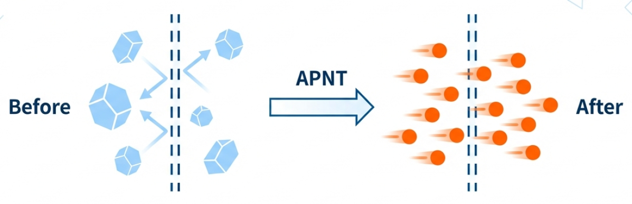
APNT® operates through a contract and co-development model. We welcome biotech and pharmaceutical companies and research institutions dedicated to new drug development to contact Formosa Pharmaceuticals to explore the infinite possibilities of technical collaboration.
ADC Programs
Formosa Pharmaceuticals’ ADC development focuses on biosimilars with fully validated market value and highly innovative bispecific ADC new drugs.
TSY-110 & TSY-120 are biosimilars for Kadcyla® and Enhertu®, respectively. Developed in alliance with EirGenix and Formosa Laboratories, these projects target HER2-driven cancers, like HER2-positive and HER2-low breast cancers. Clinical trials for TSY-110 are expected to commence in 2026.
TSY-310 is a first-in-class bispecific ADC precisely targeting EGFR and ROR1. Uniquely positioned for solid tumors such as non-small cell lung cancer (NSCLC), it possesses the potential to overcome drug resistance and address unmet medical needs in tumor therapy.

Kadcyla Biosimilar|TSY-110

TSY-110 is an ADC biosimilar targeting HER2-positive driven malignancies. By combining EirGenix’s antibodies with Formosa Laboratories’ bioconjugation platform, TSY-110 achieves high biosimilarity to the reference drug. Our goal is for TSY-110 to be the first biosimilar launched globally, providing patients and insurers with an equally effective yet more economical treatment option.
Enhertu® Biosimilar|TSY-120
TSY-120 is an ADC biosimilar referencing Enhertu®, co-developed by Formosa Pharmaceuticals and EirGenix. It targets HER2-positive and HER2-low breast cancers, aiming to be the world’s first-to-market Enhertu biosimilar.
TSY-310 | A Next-Generation Novel Bispecific ADC

TSY-310 is an innovative bispecific ADC targeting both EGFR and ROR1. With a molecular size approximately half that of traditional antibodies, it offers superior penetration. It is poised to tackle drug resistance challenges and fulfill critical unmet needs in the treatment of solid tumors like NSCLC.
Others
Anti-infective Program | TSY-210
Formosa Pharmaceuticals’ Anti-infective Program, TSY-210 possesses a broad and respected antibacterial spectrum, including nosocomial infections and resistant strains. TSY-210 addresses 9 of top 22 priority pathogens designated by WHO and CDC.