Formosa Pharmaceuticals Pipeline
TSY-310 | A Next-Generation Novel Bispecific ADC
TSY-310 is an innovative Bispecific Antibody-Drug Conjugate (ADC) development project. Engineered for dual-targeting of EGFR and ROR1, TSY-310 is positioned well for solid tumors, including Non-Small Cell Lung Cancer (NSCLC).
Precision Targeting|The EGFR & ROR1 Advantage

The therapeutic strategy of TSY-310 lies in its strategic target selection:
- EGFR: A well-known driver of solid tumor growth and metastasis, EGFR is a clinically-validated target and continues to be a critical therapeutic node across multiple cancer types.
- ROR1: A receptor protein primarily expressed during embryonic development, ROR1 is abnormally overexpressed in various cancers, including lung, breast, and hematological malignancies, making it a highly promising anti-cancer target.
By precisely co-targeting EGFR and ROR1, TSY-310 selectively eliminates tumor cells while minimizing damage to healthy tissue. This dual-action approach enhances both safety and efficacy, demonstrating sustained and stable tumor regression in solid tumor models.
Addressing the Global Burden of NSCLC and other solid tumors
Non-Small Cell Lung Cancer (NSCLC) is the primary indication for TSY-310. According to a 2022 GlobalData report, NSCLC accounts for approximately 85% of all lung cancer cases, encompassing major subtypes such as adenocarcinoma, squamous cell carcinoma, and large cell carcinoma.
The primary risk factor is aging, alongside current or former smoking, secondhand smoke exposure, and occupational exposure to carcinogens (e.g., asbestos, arsenic, chromium). Key clinical symptoms include persistent cough, chest discomfort, dyspnea, hemoptysis, unexplained weight loss, and fatigue.
Epidemiological data across 16 major markets indicate a diagnosed prevalent population of 1.5 million cases. China represents the largest market (29%), followed by the U.S. (19%) and Japan (15%). While 46 innovative therapies have reached the market, significant demand remains for late-stage clinical candidates that can overcome existing limitations.
Manufacturing and Development Partners |TSY-110 Kadcyla® Biosimilar
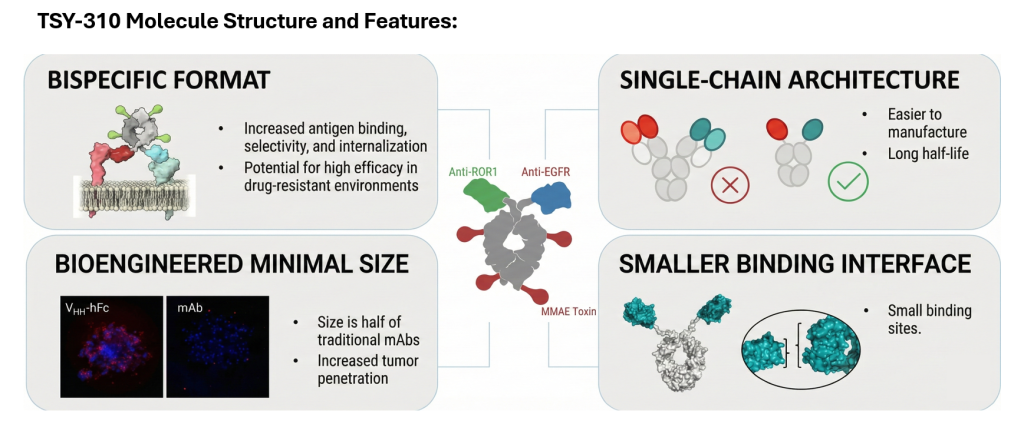
Structural Breakthroughs & Molecular Superiority
TSY-310 features a breakthrough molecular design that maximizes its therapeutic potential. Firstly, its “bispecific” design can significantly increase the binding affinity and selectivity of antigens, giving it the potential to exert high efficacy in drug-resistant environments. Secondly, TSY-310 adopts a “single-chain architecture”, simplifying the manufacturing process and maintaining a longer half-life. Furthermore, TSY-310 is only half the size of traditional monoclonal antibodies, increasing the depth and ability of the drug to penetrate tumors. Finally, the combination of a “smaller binding interface” and clinically proven MMAE toxin payload makes it more flexible and effective against solid tumors.
Compelling Preclinical Evidence
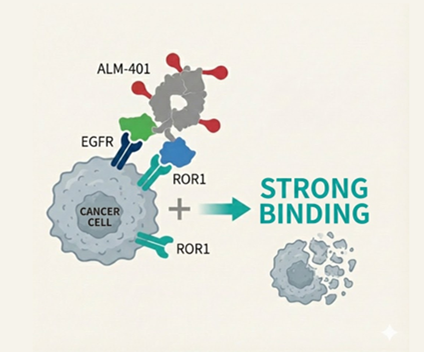
TSY-310 has demonstrated remarkable results across multiple preclinical studies:
- CDX Models: Showed high anti-tumor activity in ROR1/EGFR double-positive NSCLC cell lines. Significant regression was observed in both EGFR-mutant adenocarcinoma (PC9) and EGFR wild-type squamous NSCLC (NCI-H1703) models.
- PDX Models: TSY-310 induced substantial tumor regression in patient-derived NSCLC xenograft models.
In addition, the drug exhibits a potent bystander effect, maintaining strong activity across animal models with varying levels of target expression.
In terms of safety, TSY-310 combines both EGFR and ROR1; Since ROR1 is barely expressed in normal adult tissues, this gives TSY-310 high specificity. At the same time, the MMAEs and linkers they use have been clinically validated, and the potential risks are known and manageable.
Overcoming Resistance & Unmet Medical Needs
Drug resistance remains the greatest hurdle in oncology. TSY-310 is positioned as a potential solution to this challenge.
Existing EGFR-targeted therapies (1st to 3rd generation TKIs and mAbs) inevitably lead to resistance.
Research indicates that ROR1 expression often increases when cancer cells develop resistance to EGFR inhibitors. TSY-310 exploits this vulnerability by actively identifying and attacking these resistant cells, addressing a critical unmet medical need where current therapies fail.
Outlook: A High-Value Clinical Asset
The ADC landscape is currently one of the most active sectors for pharmaceutical licensing. Since 2024, multiple ADC deals have exceeded the $1 billion mark. As there are currently no approved Bispecific ADCs on the market, TSY-310 represents a rare and innovative asset with significant clinical potential and commercial value.